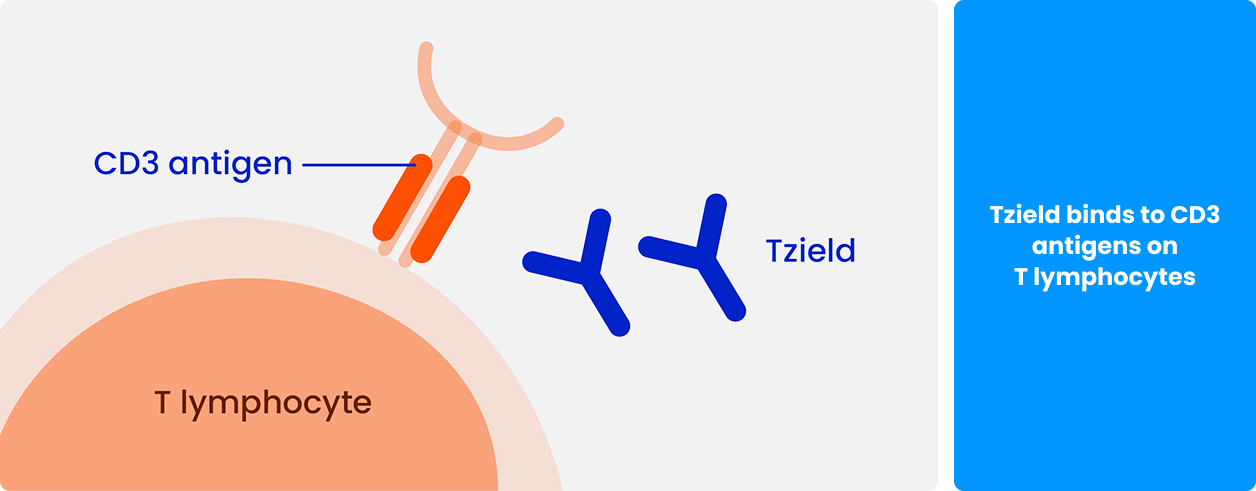
How does Tzield (teplizumab) work?
Tzield is a medicine that contains the active substance teplizumab. It belongs to a group of medicines called ‘monoclonal antibodies'.1,2
Tzield is a protein that recognises and sticks to a target. The target is a substance called CD3 that is found on a type of white blood cell – called ‘T lymphocytes’.1,2
When Tzield sticks to T lymphocytes it can help to slow down the attack on beta cells.2,3
By slowing down the autoimmune attack on beta cells, your pancreas can keep producing insulin on its own for
a little longer.2,3
Tzield is a medicine used to delay the start of Stage 3 Type 1 diabetes. This condition happens when the body cannot make enough insulin on its own and may need insulin injections. This medicine is used in adults and children aged 8 years and older who have Stage 2 Type 1 diabetes.1
If you have any questions on how Tzield works or about your treatment with Tzield, ask your doctor.
This website is not intended to be a substitute for medical advice. Please refer to the Patient information leaflet and the Patient Guide for full safety information. You may have received these items from your doctor already.
The contents of this website have been developed and funded by Sanofi.
References:
- TZIELD® (teplizumab) UK Patient Information Leaflet. Accessed: November 2025.
- TZIELD® (teplizumab) UK Summary of Product Characteristics. Accessed: November 2025.
- Nagy G, Szekely TE, Somogyi A, et al. New therapeutic approaches for type 1 diabetes: Disease-modifying therapies. World J Diabetes. 2022; 13(10): 835–850.
Reporting side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in the package leaflet. You can also report side effects directly via the Yellow Card Scheme at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple app Store.
Alternatively, you can report side effects to Sanofi on Tel: 0800 0902 314 or send via email to UK-drugsafety@sanofi.com. By reporting side effects, you can help provide more information on the safety of this medicine.
▼This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects that you may get. See www.mhra.gov.uk/yellowcard for how to report side effects.
© 2025 Sanofi. All rights reserved. MAT-XU-2501102 (V1.0) | November 2025